How AI in Healthcare Actually Works: Behind-the-Scenes Technology
The transformation of medical practice through artificial intelligence represents one of the most significant technological shifts in modern medicine, yet the actual mechanisms driving these innovations remain largely invisible to most stakeholders. Understanding how these systems function at a technical level reveals both their tremendous potential and inherent limitations. The infrastructure supporting intelligent diagnostic tools, predictive analytics platforms, and automated treatment protocols operates through sophisticated combinations of machine learning architectures, data processing pipelines, and clinical integration frameworks that work seamlessly behind the interface healthcare professionals interact with daily.
The fundamental operations powering AI in Healthcare begin with data ingestion processes that aggregate information from electronic health records, medical imaging systems, laboratory information management platforms, and continuous monitoring devices. These diverse data streams undergo extensive preprocessing through normalization algorithms that standardize formats, handle missing values, and ensure interoperability across systems developed by different vendors using incompatible standards. The quality of this foundational data processing directly determines the reliability of every downstream analytical function, making it the most critical yet underappreciated component of healthcare AI infrastructure.
Neural Network Architectures in Medical Imaging Analysis
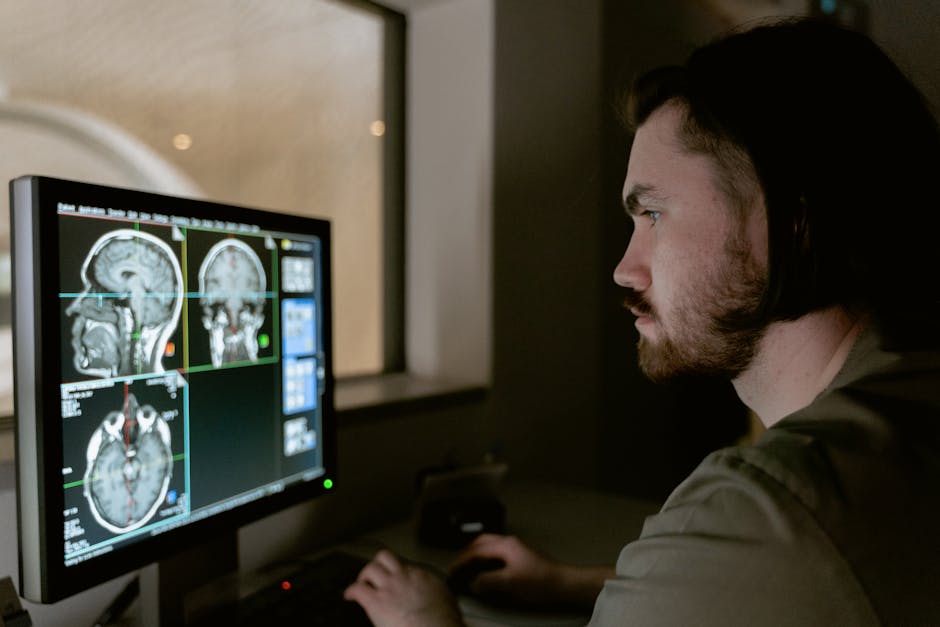
Medical imaging applications of AI in Healthcare rely predominantly on convolutional neural networks specifically adapted for volumetric data processing. These architectures process three-dimensional medical scans through successive layers of mathematical operations that progressively extract higher-level features from raw pixel intensities. The initial convolutional layers identify basic patterns like edges and textures, while deeper layers recognize complex anatomical structures and pathological indicators. Transfer learning techniques allow these networks to leverage knowledge gained from analyzing millions of general images before specialization on medical datasets, dramatically reducing the training data requirements that would otherwise make clinical implementation impractical.
The actual inference process when a radiologist submits a CT scan for AI analysis involves several computational stages. First, the volumetric data undergoes spatial normalization to standard anatomical coordinates, ensuring consistent orientation regardless of how the scan was acquired. Attention mechanisms then guide the network to focus computational resources on regions with higher diagnostic uncertainty, mimicking how human radiologists systematically examine images. The final classification layers produce probability distributions across diagnostic categories along with spatial heatmaps indicating which image regions most influenced each conclusion, providing the interpretability healthcare providers require before trusting algorithmic recommendations.
Natural Language Processing for Clinical Documentation
Healthcare Technology employing natural language processing tackles the enormous volume of unstructured clinical notes, discharge summaries, and pathology reports that contain diagnostic insights not captured in coded data fields. Modern transformer-based language models process these documents through self-attention mechanisms that evaluate relationships between all words simultaneously rather than sequentially, capturing long-range dependencies crucial for understanding complex medical narratives. Domain-specific pretraining on medical literature and clinical notes teaches these models the semantic patterns of medical language before fine-tuning on specific tasks like symptom extraction, disease classification, or treatment recommendation.
The technical implementation of clinical NLP systems faces unique challenges absent from general language processing applications. Medical terminology exhibits extreme variability with abbreviations, synonyms, and context-dependent meanings that generic language models handle poorly. Named entity recognition components must distinguish between mentions of patient conditions, family medical histories, and hypothetical scenarios discussed during differential diagnosis. Negation detection algorithms determine whether documented symptoms are present or explicitly ruled out, a distinction that fundamentally changes clinical interpretation. Temporal reasoning modules establish chronological relationships between events mentioned across multiple documents created over months or years of treatment.
Integration with Clinical Workflows
The practical deployment of Medical AI Applications requires sophisticated middleware that bridges machine learning systems with clinical information systems while preserving existing workflows. HL7 FHIR interfaces enable standardized data exchange between AI platforms and hospital systems, automatically retrieving relevant patient information when needed and returning algorithmic insights in formats clinicians can immediately use. Real-time inference engines must deliver results within milliseconds to support point-of-care decision-making without introducing delays that disrupt patient care. Fallback mechanisms ensure graceful degradation when AI systems encounter edge cases outside their training distribution, routing such cases to human experts rather than producing unreliable outputs.
Predictive Analytics and Risk Stratification Systems
Population health management applications of AI in Healthcare employ gradient boosting machines and ensemble methods to identify patients at elevated risk for adverse outcomes like hospital readmission, disease progression, or medication non-adherence. These models process hundreds of features spanning demographic information, clinical measurements, medication histories, social determinants of health, and behavioral indicators to produce individualized risk scores. The training process involves carefully constructed validation strategies that account for temporal dynamics, ensuring models evaluated on historical data will generalize to future patients rather than simply memorizing patterns specific to the training period.
Behind the risk scores presented to care coordinators operates a complex feature engineering pipeline that transforms raw clinical data into predictive signals. Time-series analysis components extract trends and variability patterns from longitudinal measurements like blood pressure or glucose levels. Medication interaction analyzers evaluate polypharmacy risks by modeling combinatorial effects of current prescriptions. Social network features quantify care fragmentation by analyzing patterns in provider visits and care transitions. Missing data imputation algorithms fill gaps using learned relationships between observed variables, crucial given the sparse and irregular nature of real-world clinical data collection.
Continuous Learning and Model Updating Frameworks
Healthcare AI systems require ongoing adaptation as medical knowledge evolves, treatment protocols change, and patient populations shift. Active learning frameworks identify cases where model uncertainty exceeds acceptable thresholds, routing these examples to human experts whose feedback continuously refines the training dataset. Federated learning architectures enable model improvement across multiple healthcare institutions without centralizing sensitive patient data, instead distributing model training across local datasets and aggregating only the learned parameters. Drift detection algorithms monitor incoming data distributions and model performance metrics to identify when retraining becomes necessary, preventing silent degradation as real-world conditions diverge from training assumptions.
The technical infrastructure supporting model updates includes versioning systems that track every deployed model iteration along with the specific training data, hyperparameters, and validation metrics associated with each version. A/B testing frameworks enable controlled deployment where new model versions serve a subset of requests while performance metrics are compared against established baselines before full rollout. Rollback mechanisms allow instant reversion to previous versions if monitoring systems detect unexpected behavior in production. This software engineering discipline around model lifecycle management distinguishes production healthcare AI systems from research prototypes, ensuring the reliability required for clinical applications.
Privacy-Preserving Computation Techniques
Implementing AI in Healthcare while protecting patient privacy requires specialized cryptographic protocols that enable computation on encrypted data. Homomorphic encryption schemes allow mathematical operations on encrypted medical records without decryption, producing encrypted results that only authorized parties can interpret. Secure multi-party computation protocols distribute sensitive computations across multiple servers such that no single entity accesses complete patient information, yet correct algorithmic outputs emerge from the collaborative process. Differential privacy mechanisms inject carefully calibrated noise into training datasets and model outputs, providing mathematical guarantees that individual patient information cannot be reverse-engineered from published models or aggregate statistics.
The practical implementation of these privacy-preserving techniques involves substantial computational overhead compared to standard machine learning pipelines. Encrypted operations execute orders of magnitude slower than their plaintext equivalents, requiring specialized hardware acceleration and algorithmic optimizations to achieve acceptable performance. Noise injection for differential privacy creates inherent accuracy-privacy tradeoffs that must be carefully balanced based on application sensitivity and regulatory requirements. Despite these challenges, privacy-enhancing technologies increasingly enable collaborative research and model development across institutions that could not share raw data under existing regulations, unlocking training datasets large enough to support robust Medical AI Applications.
Conclusion
The sophisticated technical infrastructure enabling AI in Healthcare extends far beyond the user interfaces clinicians interact with, encompassing complex data processing pipelines, specialized neural architectures, privacy-preserving computation frameworks, and rigorous model governance systems. Understanding these underlying mechanisms provides essential context for evaluating algorithmic capabilities and limitations, setting appropriate expectations for clinical deployment, and identifying areas requiring continued research and development. As healthcare organizations increasingly invest in intelligent systems, similar technological transformations are reshaping other sectors, with AI Banking Solutions demonstrating how these foundational technologies adapt across industries to address sector-specific challenges while maintaining the core principles of data-driven decision support and automated intelligence.



Comments
Post a Comment